Clinical trials
Observational research (NIS)
PASS research
Medical registers
PMS Study
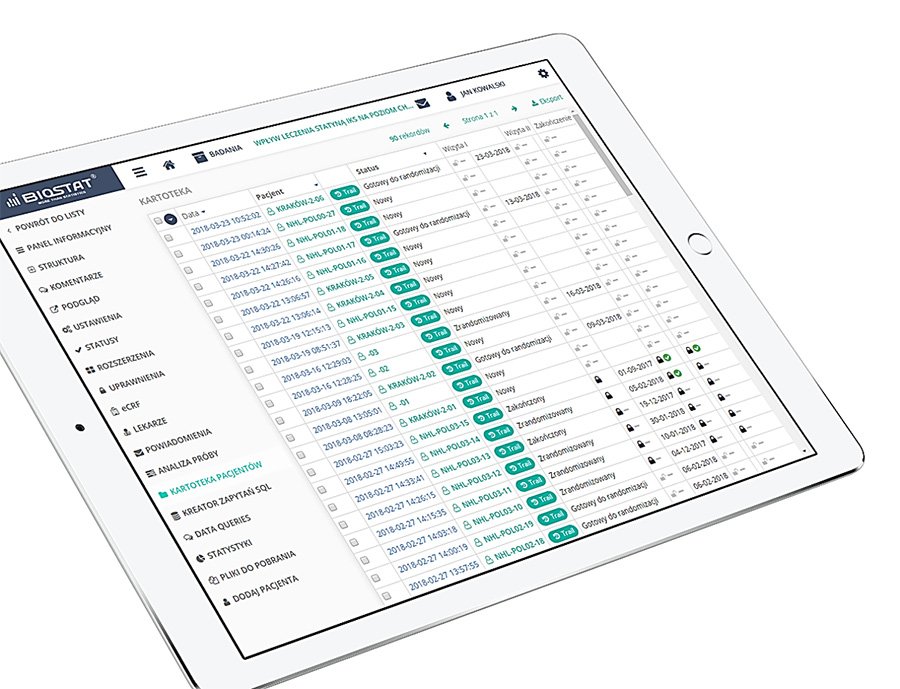
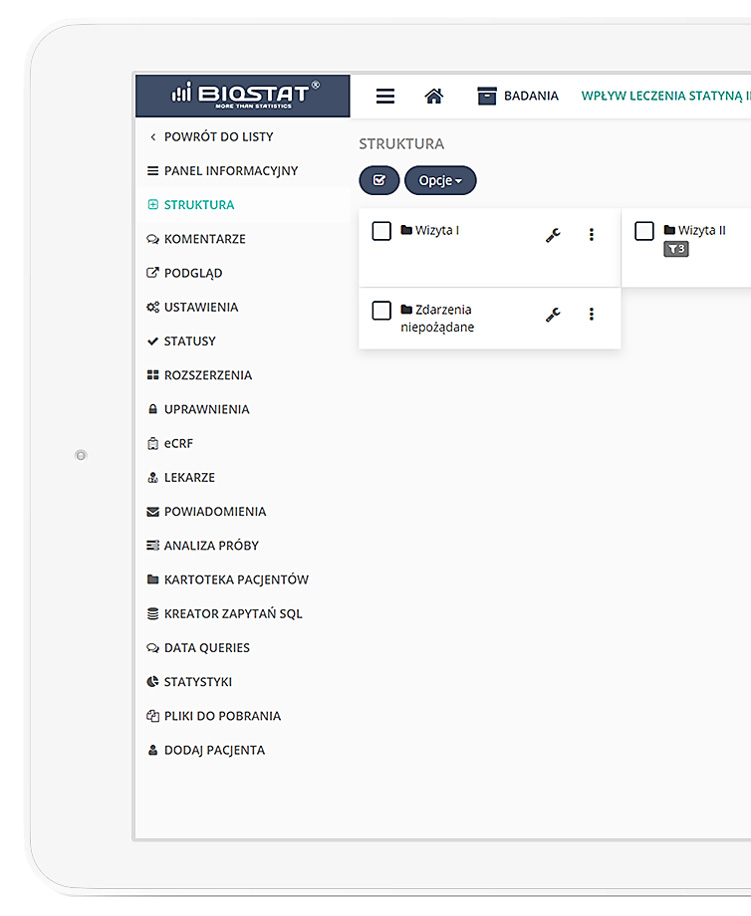
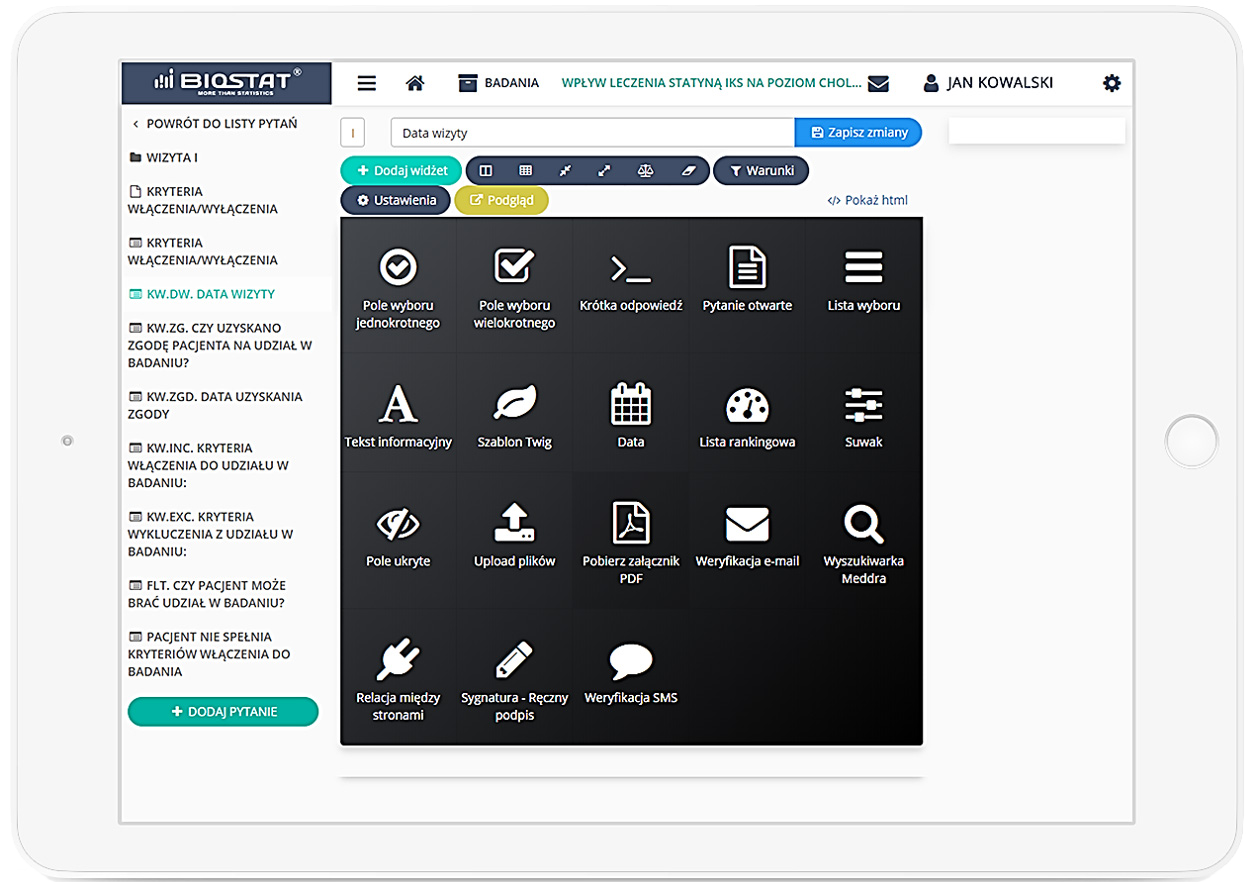
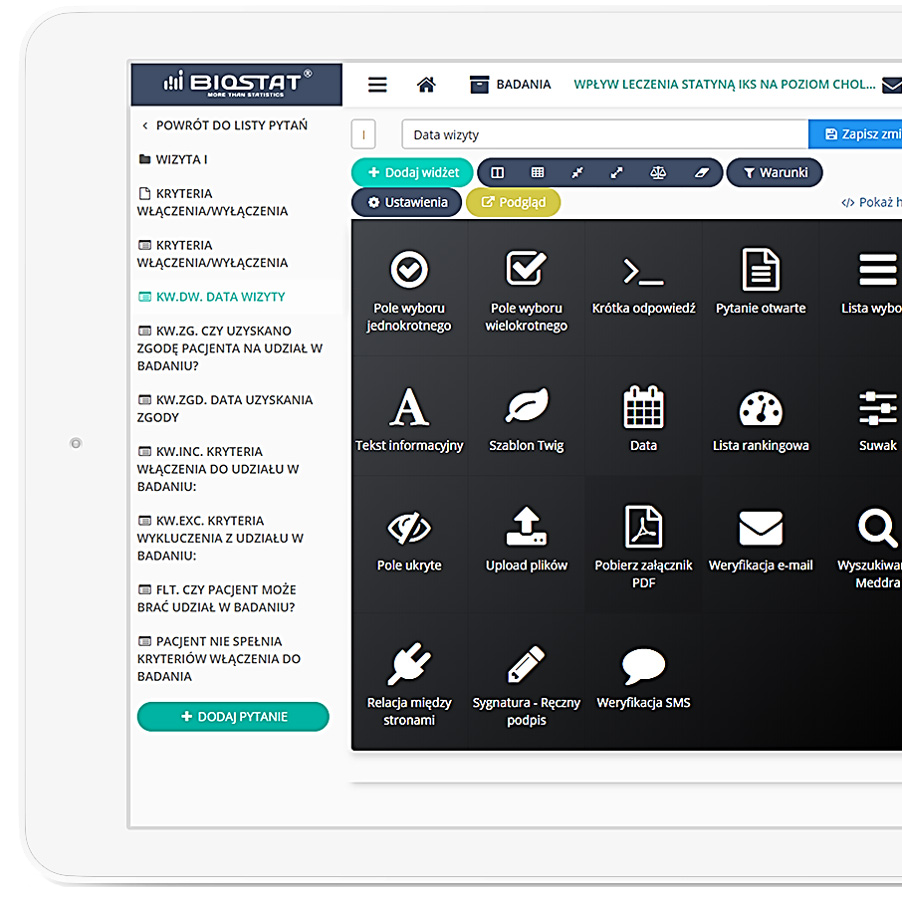
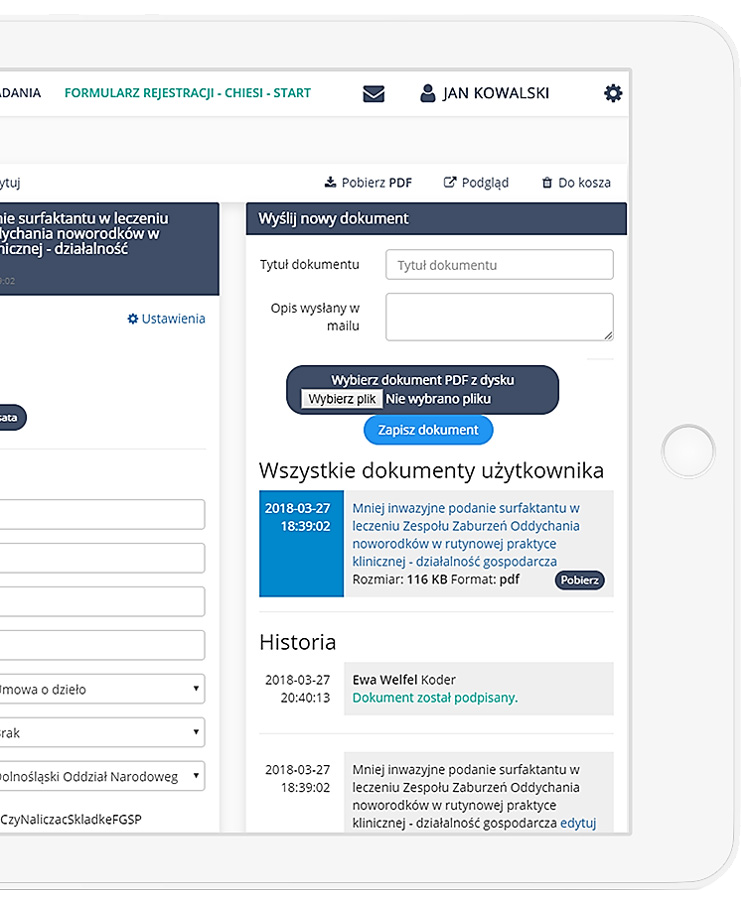
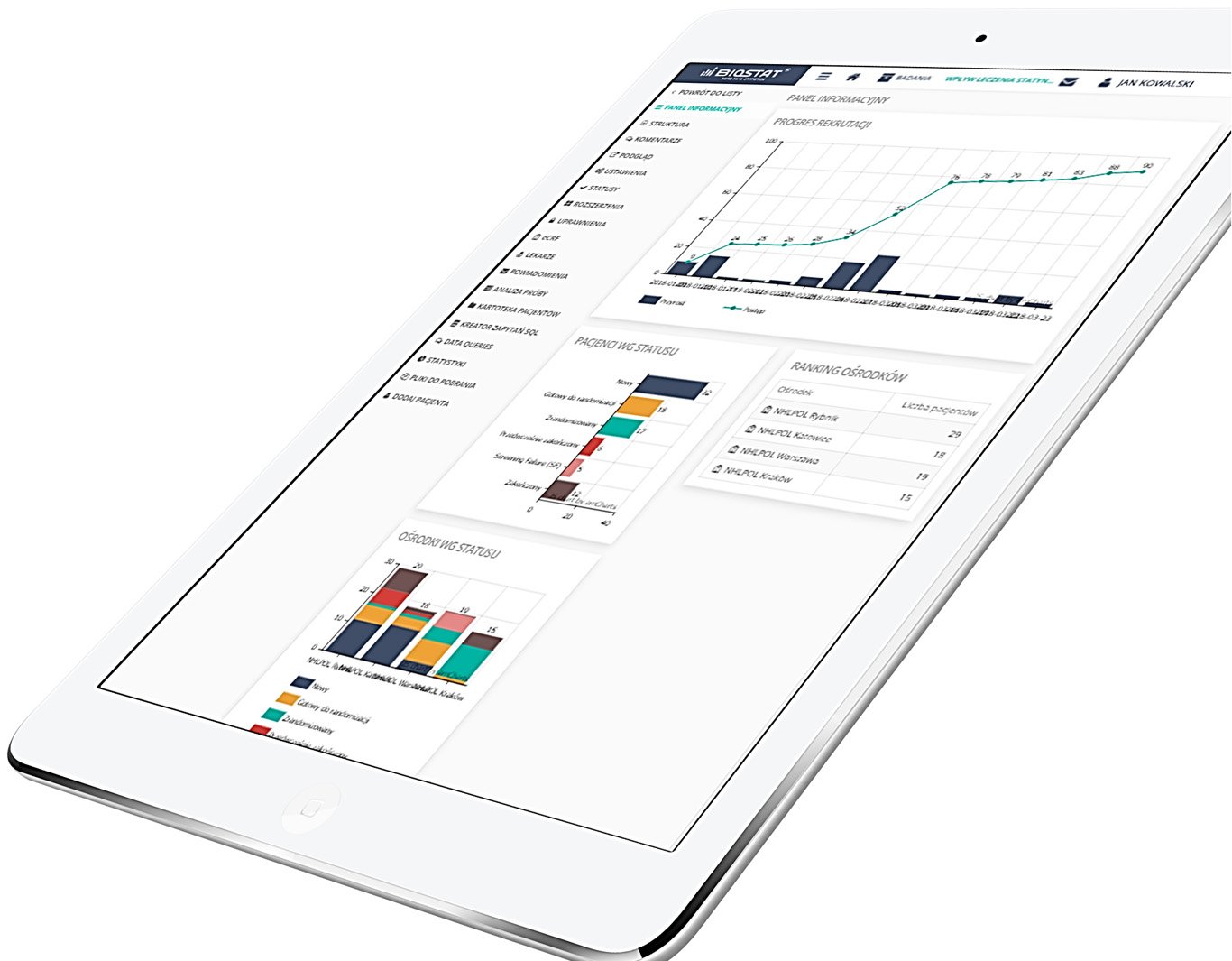
eCRF.bizTM
Move your research
Into a digital world
The system has been created by professionalists for professionalists in accordance with FDA Title 21 CFR Part 11 standards.
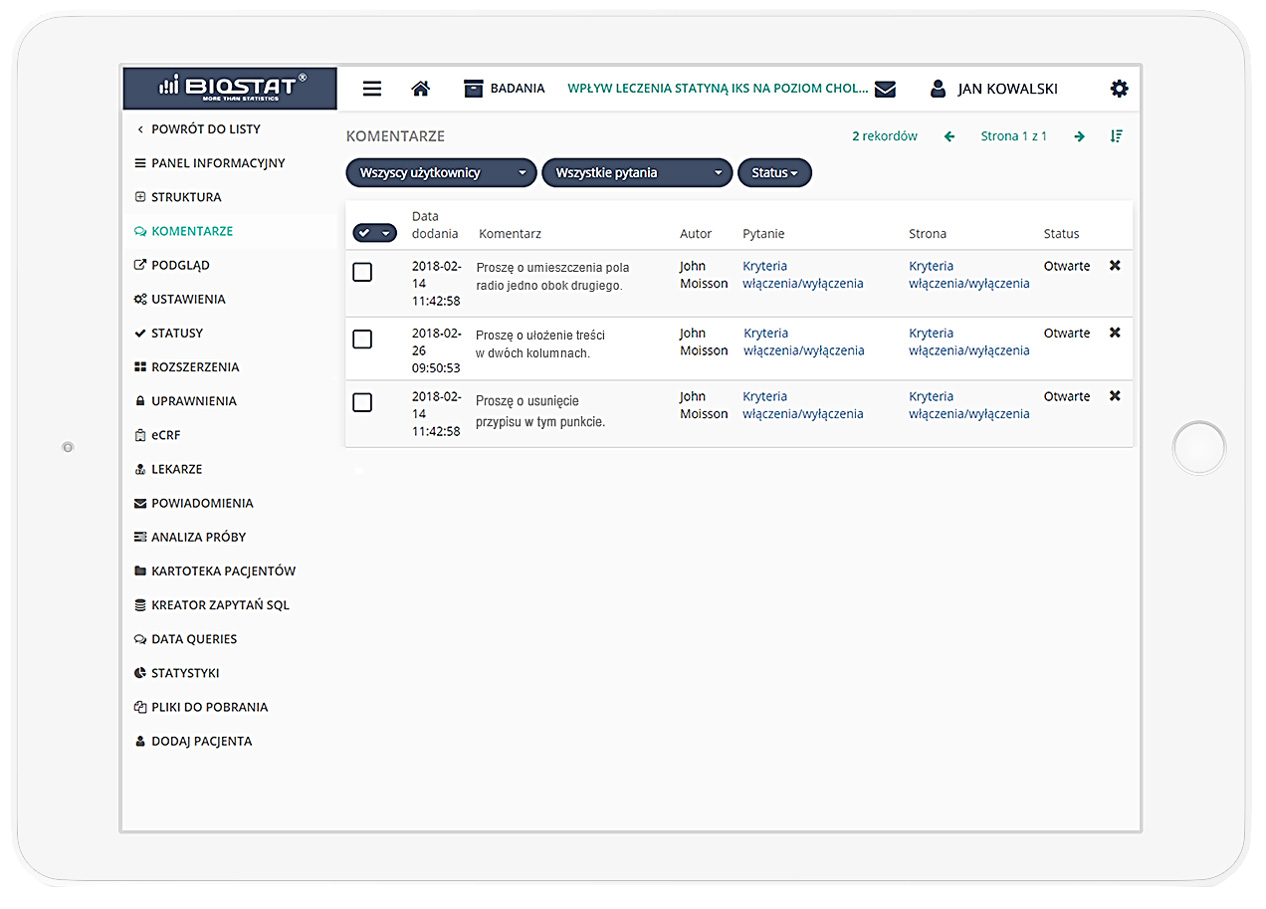
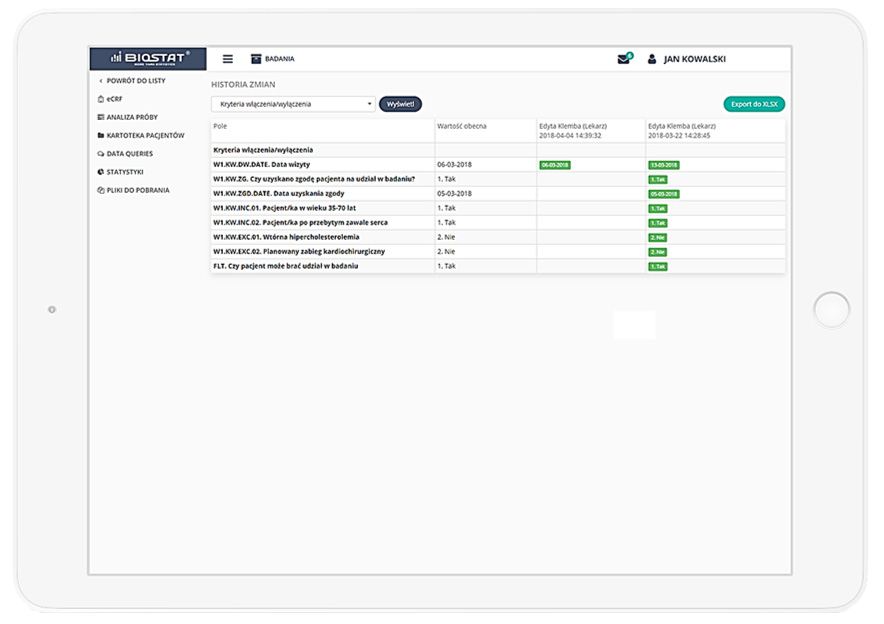
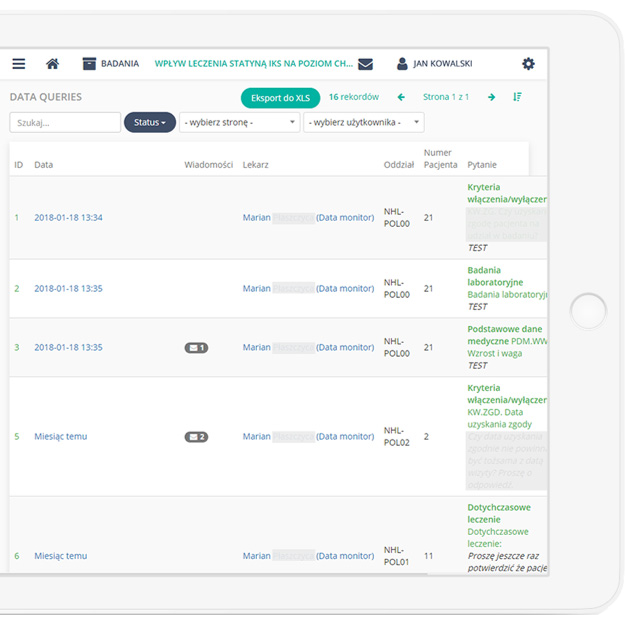
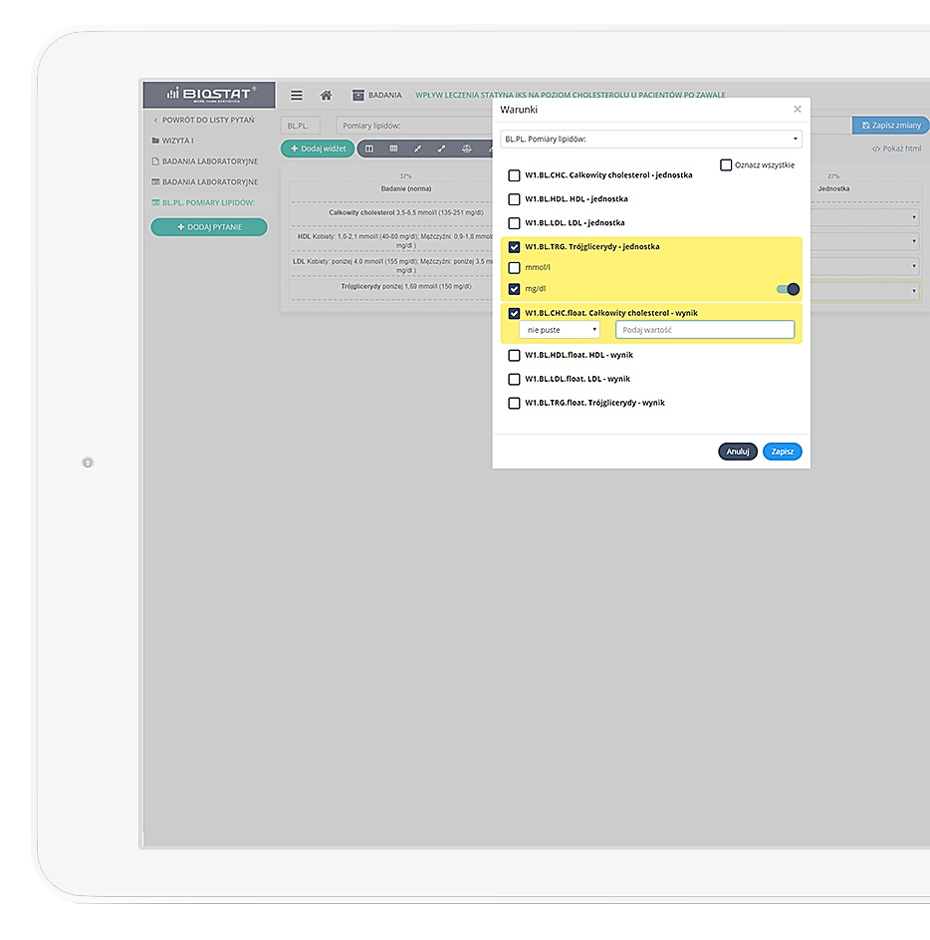
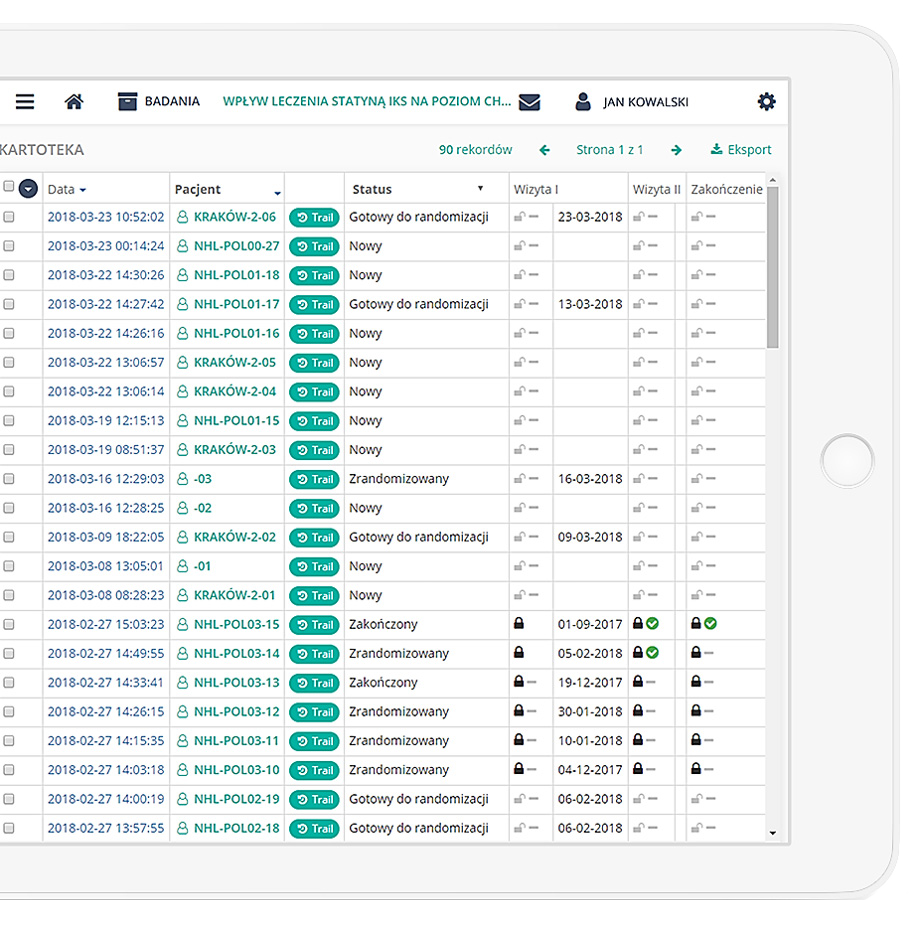
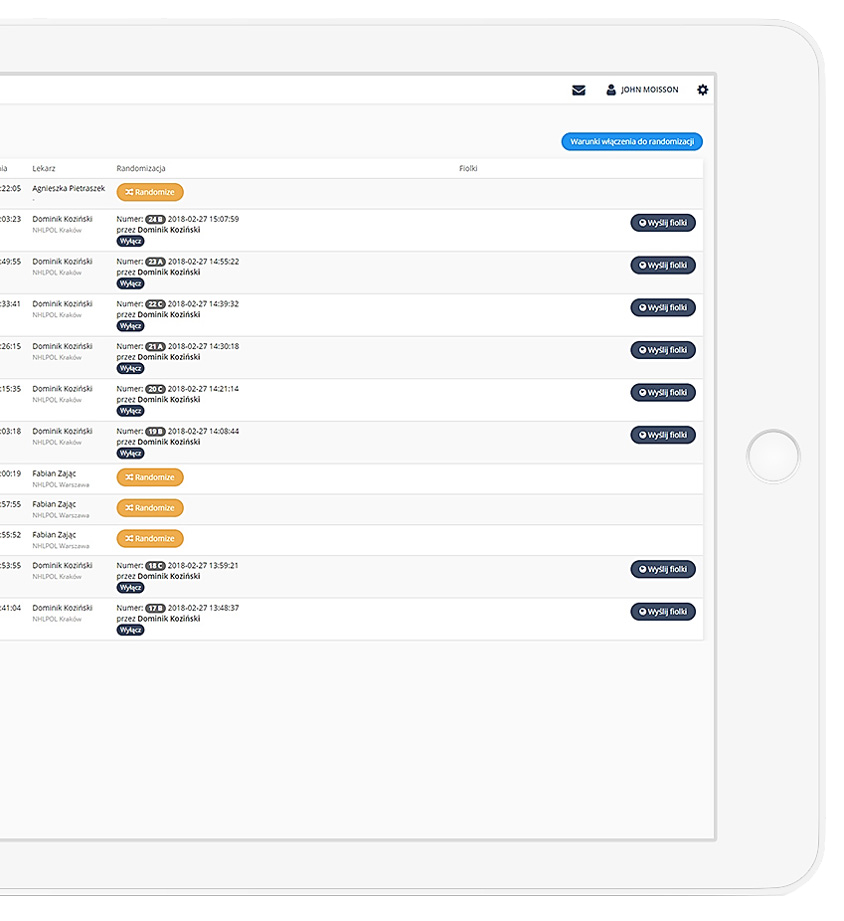
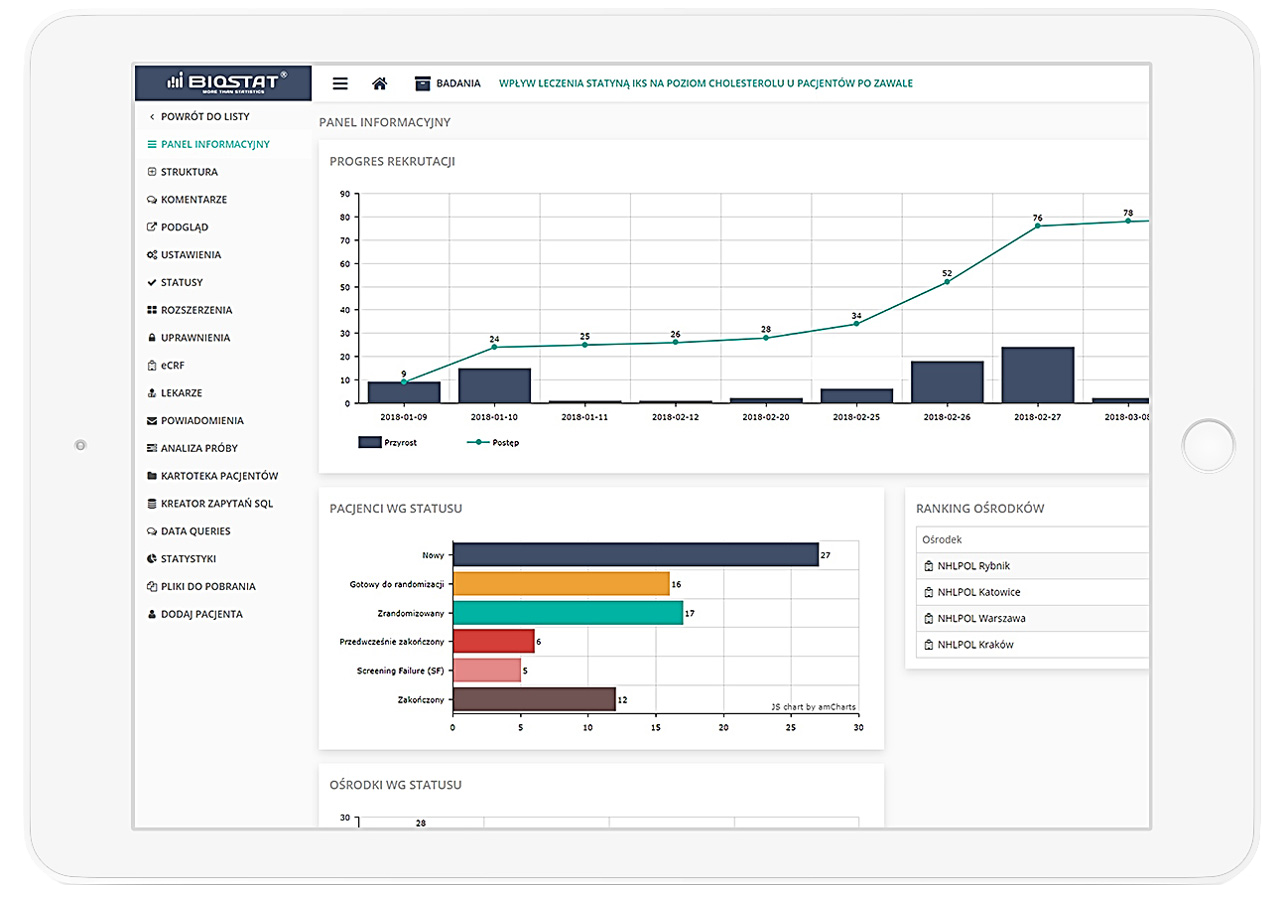
Precise data management and their completeness.
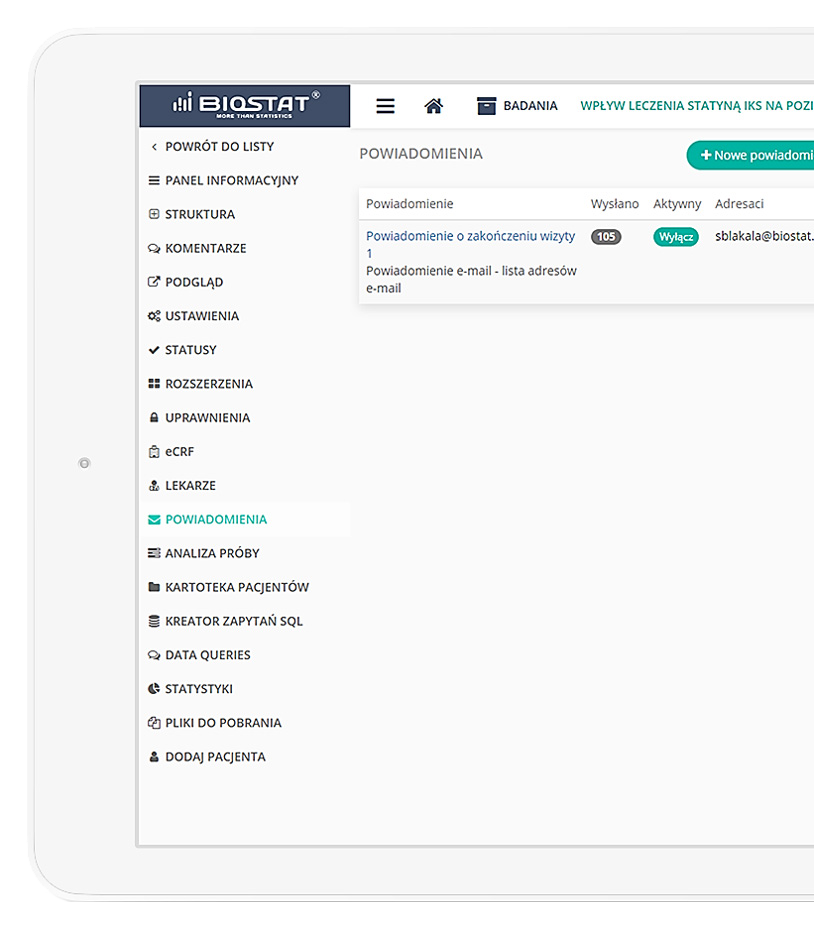
IT team, statisticians, and in-house data managers.
Precise data management and their completeness.
IT team, statisticians, and in-house data managers.