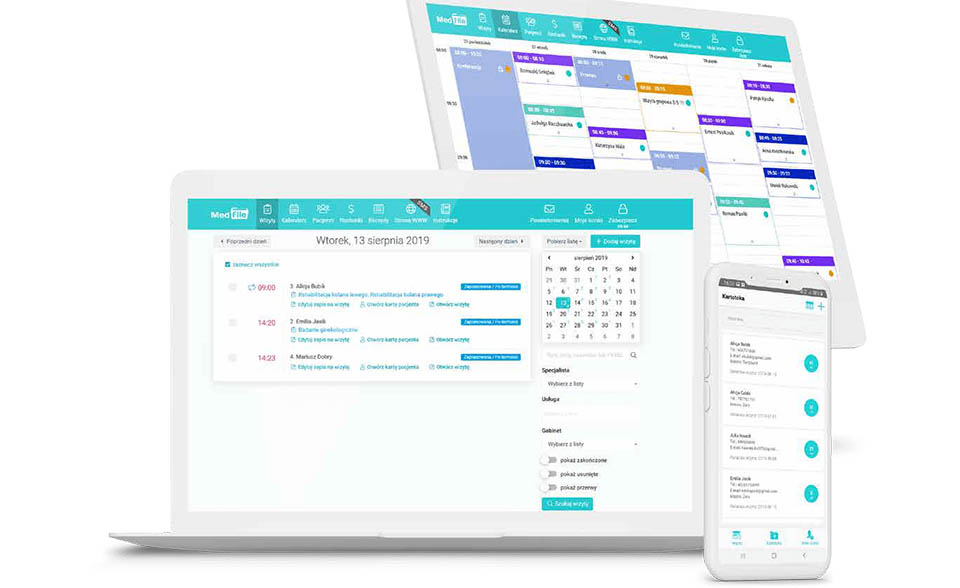
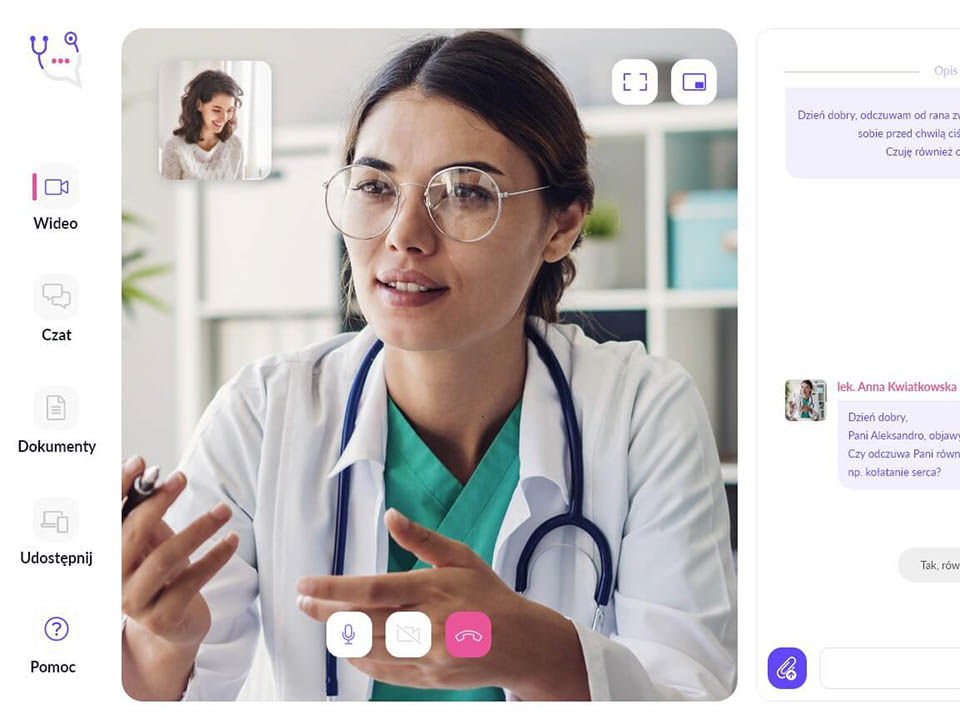
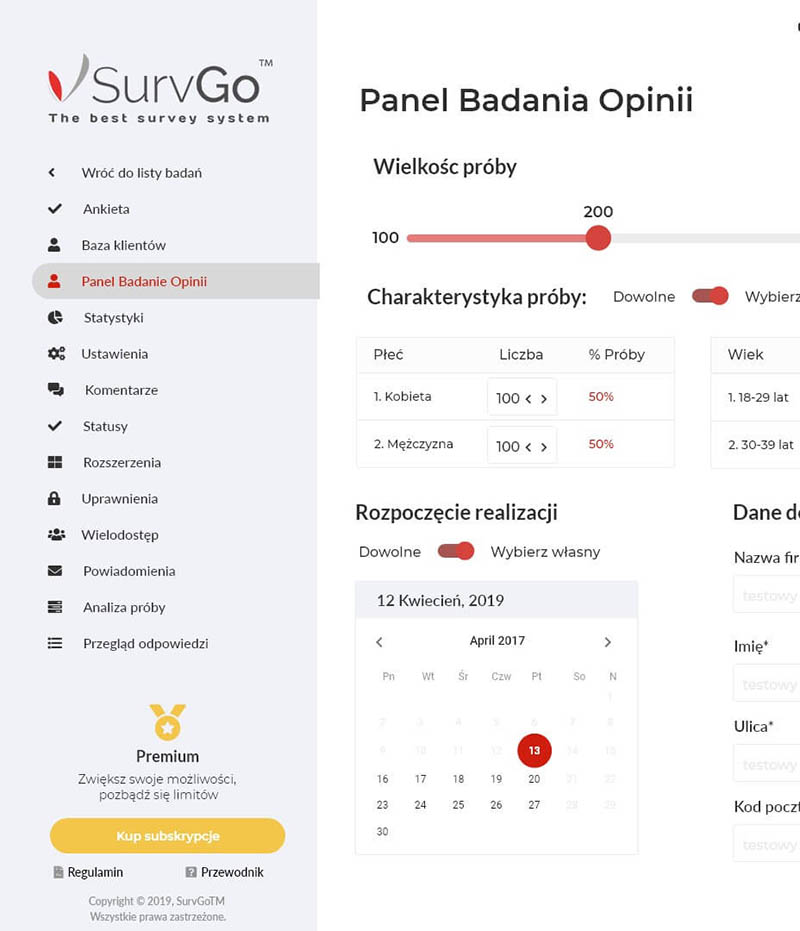
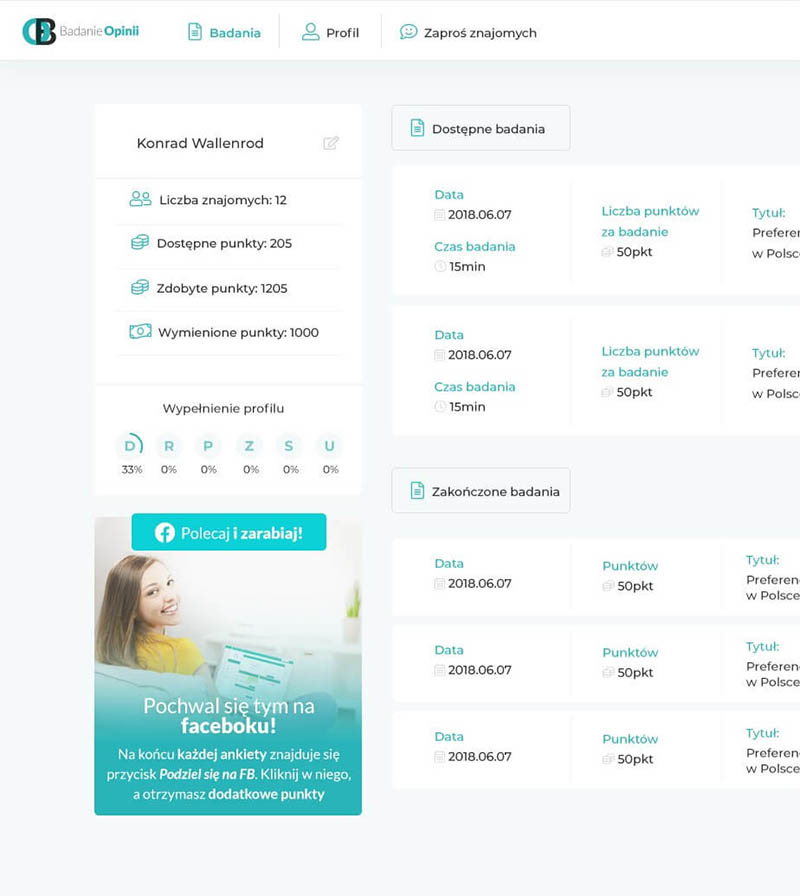
Leader on the Polish market in providing professional and innovative solutions in the area of clinical trials and observational studies, statistics, medical software, telemedicine and R&D.
Commercial research unit with status of a Research and Development Centre (R&DC) granted by the Minister of Entrepreneurship and Technology.